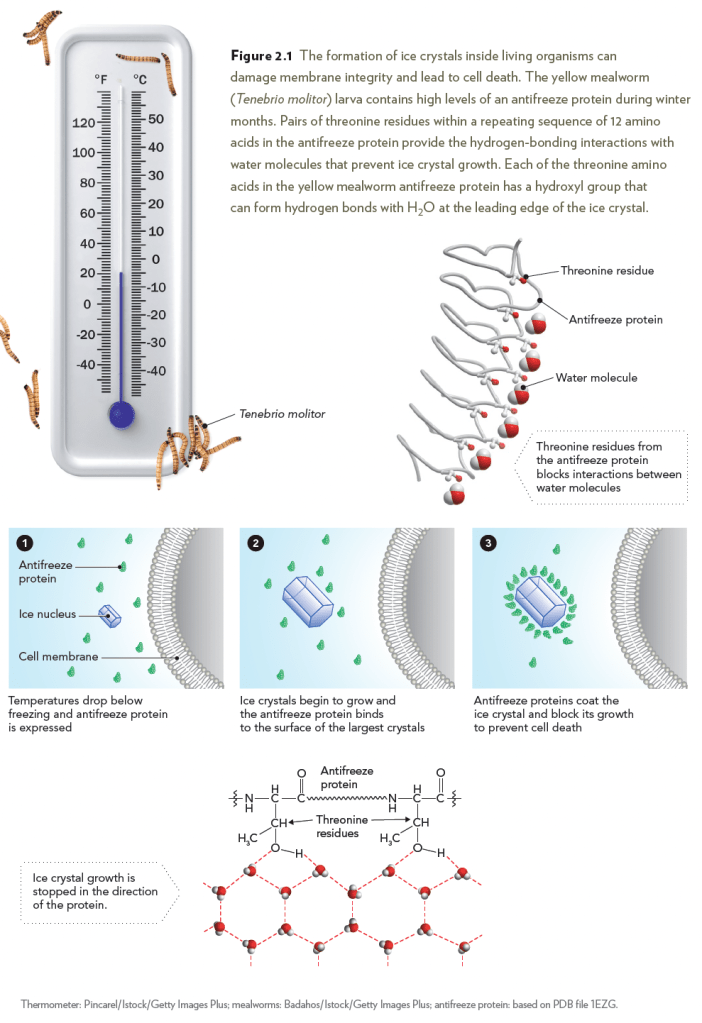
Water is critical for life and is considered the universal solvent because
of its unique properties. When temperatures drop below the freezing
point of water, however, ice crystals form and can be lethal to cells. Cold-blooded
organisms that live in cold climates have evolved mechanisms
to cope with freezing temperatures, one of which is the synthesis of antifreeze
proteins that block ice crystal growth. The larval antifreeze protein
contains multiple stretches of 12 amino acids that repeat the sequence
threonine–cysteine–threonine. The hydroxyl groups on threonine residues
within the repeated amino acid sequence are located at precisely the
spacing needed to maximize hydrogen-bond formation with water molecules,
thereby blocking further growth of the crystal lattice.