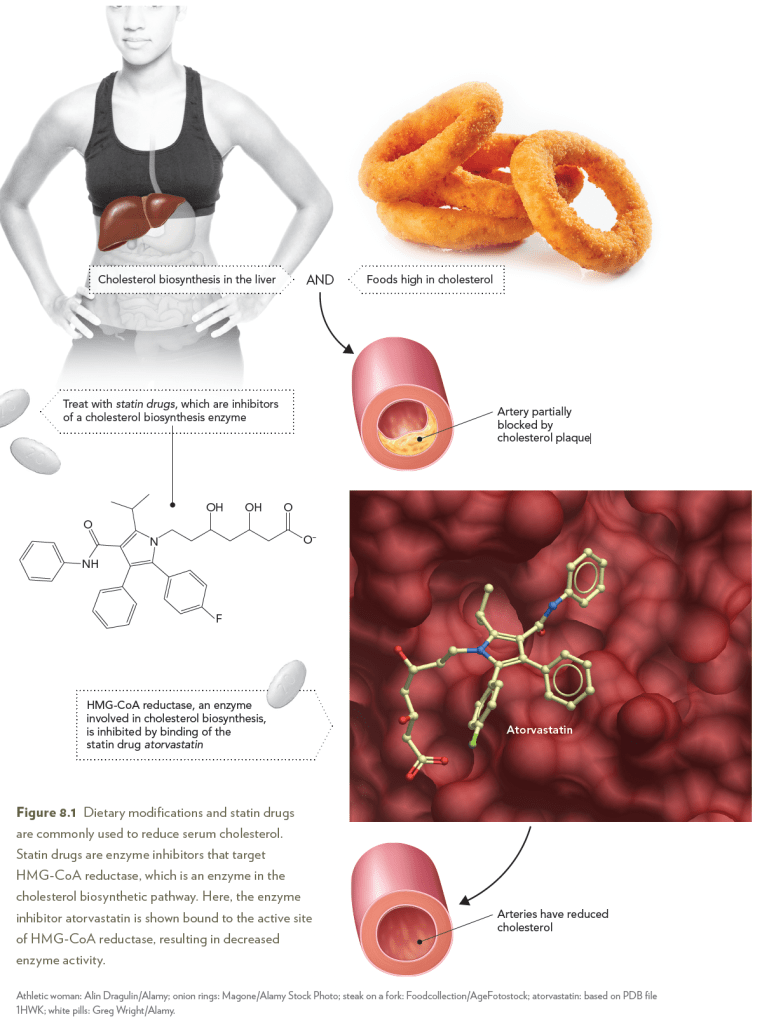
High serum cholesterol is associated with heart disease and has been
attributed to diets that are rich in cholesterol-containing foods and to
elevated synthesis of cholesterol in the liver. Inhibition of the enzyme
3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase lowers
intracellular cholesterol levels by reducing the rate of cholesterol synthesis
in the liver. Importantly, when intracellular cholesterol is lowered
by inhibition of HMG-CoA reductase, liver cells remove more cholesterol
from the serum, thereby decreasing circulating cholesterol levels
and reducing the risk of heart disease. Structural, kinetic, and mechanistic
studies of HMG-CoA reductase in the 1970s paved the way for the
development of a large class of HMG-CoA reductase inhibitors called
statin drugs that are in widespread clinical use today. With detailed information
about how inhibitors bind to enzymes and block their activity,
new drugs with improved inhibitory actions can be designed.